Contact your provider
This article was updated May 23, 2022, to reflect new information from the CDC.
When it comes to getting a vaccine to protect yourself against a disease like COVID-19, booster shots can be a normal part of the vaccination process.
“Booster” basically refers to an extra dose of a vaccine after the original (or primary) dose is administered. These boosters can be weeks, months or years later, according to Sandy Salverson, PharmD, vice president of Pharmacy Operations at OSF HealthCare.
“Most adults have a memory of having to get a booster for a childhood disease, such as measles, whooping cough or meningitis. Or for a disease like tetanus, where booster shots are recommended every 10 years as the original immunity wanes,” Sandy said.
What do COVID-19 booster shots do?
Some vaccines are administered using the strategy of giving the original (or primary) dose along with a booster dose. With the original dose, the immune system is primed to recognize and produce antibodies against the virus it was created to defend.
Booster shots are administered to remind the body’s immune system about the virus it needs to defend against. This improves or gives the immune system a boost.
Extra doses for immunocompromised
It’s recommended by the U.S. Centers for Disease Control and Prevention that moderately to severely immunocompromised individuals age 5 and older who received the two-dose series of either Pfizer or Moderna get a third dose of the vaccine four weeks following their second primary dose. It’s further recommended that individuals age 12 and older receive a fourth dose at least three months after their third dose.
Children age 5 to 17 who are immunocompromised can only receive Pfizer. It is not recommended that children age 5 to 11 receive a fourth dose (a second booster).
Adults age 18 and older who are immunocompromised and received the one-dose Johnson & Johnson vaccine, should get a booster two months after their primary dose. The CDC further recommends that these individuals get a second booster at least four months after their first booster, but it should be an mRNA vaccine – Pfizer or Moderna.
“A booster is given when a person has completed their vaccine series and the immunity protection against the virus begins to decrease with time, as data has shown,” Sandy said. “The additional doses are strictly to improve the immunity response of those moderately to severely immunocompromised individuals.”
These individuals are especially vulnerable to COVID-19 and are more at risk of serious, prolonged illness. Their level of immunity following the original doses of the vaccine is lower than people who are not immunocompromised, Sandy said.
“This isn’t because the vaccine isn’t effective. It’s because the immune systems of these individuals need the additional dose or doses as part of their primary series to reach the level of immunity that best protects them from severe illness or hospitalization,” Sandy said.
COVID-19 boosters
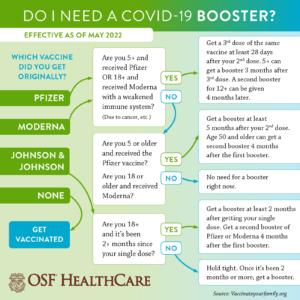
Do I need a booster?
Download
The Federal Drug Administration (FDA) and the CDC have approved all three vaccines available – Moderna, Pfizer and Johnson & Johnson – for use as a booster in people age 18 and older. The agencies have approved Pfizer as a booster for kids over age 5.
Guidance calls for Moderna or Pfizer COVID-19 booster shots to be five months after the primary two-dose series. Further, individuals age 50 and older can receive a second booster of Pfizer or Moderna at least four months following their first booster shot.
As for Johnson & Johnson, the recommendation is those age 18 and older who received the one-dose vaccine get a booster two months later. It is further recommended those individuals get a second booster of an mRNA vaccine – Pfizer or Moderna – four months after their first booster.
“Booster doses are meant to remind the immune system how to respond to viruses or bacteria, so a response can be hard wired,” Sandy said. “Studies have shown us that once the immune system is primed, additional doses of the COVID-19 vaccines will help finalize the memory within the immune system.”
Schedule your appointment
If you qualify for a booster, or are unsure if you qualify, contact your primary care provider or schedule an appointment through OSF MyChart.
Last Updated: February 16, 2024
