How vaccines work to protect us
Are your vaccines up-to-date?
Vaccines are one of the most effective tools we have to prevent serious illness. They train the body’s natural defenses to recognize harmful bacteria or viruses, so your immune system is ready if you’re exposed later. By doing this, vaccines protect both individuals and communities from dangerous – sometimes even deadly – diseases.
But how do vaccines work? And how does getting vaccinated protect others through herd immunity?
How the Body Fights Infection
“Our bodies are made up of trillions of cells, each with a purpose. While still working on the total, scientists estimate the number of cells at around 30 trillion,” Brian Curtis, MD, vice president, chief medical officer, OSF Medical Group, said. “When a bacteria or virus invades the body, the intruder begins to multiply and attack or mimic our cells, infecting us and causing us to become ill.”
Our immune system has several ways to fight the intruders. While our blood carries oxygen to tissues and organs, it also contains white blood cells, or immune cells, that fight infection. They recognize invaders, attack them and help us recover.
Three main types of cells play a role:
- Macrophages: Swallow and digest harmful germs, leaving behind pieces called antigens
- B-lymphocytes: Produce antibodies that attach to antigens and mark them for destruction
- T-lymphocytes: Destroy infected cells directly
This process can take several days, which is why you may feel sick while your body builds a defense.
“After we defeat the infection, our bodies often catalog the invader and remember how to protect us against that disease in the future,” Dr. Curtis said. “It’s a constant battle worthy of Hollywood’s biggest screen.”
When you get a vaccine, you build up vaccine immunity. When you get a vaccine, you build up vaccine immunity, which helps protect you and your loved ones from severe symptoms.
What do vaccines do?
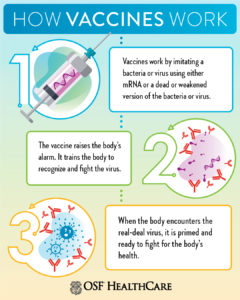
Vaccines give your immune system a “practice run” without making you seriously ill. They imitate an infection by introducing a dead, weakened or harmless piece of a germ, which your body learns to recognize and protect against. After getting vaccinated, if you are exposed to the germ later, your body already has the knowledge to fight the infection.
Vaccines:
- Teach your immune system to fight specific diseases
- Help prevent dangerous infections before they start
- Provide protection for you and those around you
This is the key difference in the conversation of vaccination vs. immunization. Vaccination is the act of getting the shot. Immunization is the process that happens afterward – when your body builds lasting protection.
As is the case in traditional vaccines, our immune system remembers the germ and is more prepared to attack it should we become infected for real. The imitation infection caused by a vaccine may cause minor symptoms, such as fever. You should expect these minor symptoms as your body builds immunity.
“Once your body makes the required antibodies, your body remembers how to fight that disease in the future,” Dr. Curtis said. “Because it takes a couple of days to weeks for our bodies to build up an immunity, it’s still possible to get infected by the targeted disease just before or just after the vaccination.”
How do mRNA vaccines work?
Traditional vaccines use dead or weakened germs. A newer method – mRNA vaccines – takes a different approach.
For example, COVID-19 mRNA vaccines contain instructions that tell your cells how to make a harmless piece of the virus’s spike protein. Once your body sees this protein, your immune system treats it as an invader and starts building antibodies.
- The mRNA never enters the nucleus of the cell, so it does not affect or change your DNA.
- After making the protein piece, your body destroys the mRNA instructions.
- The immune system remembers how to fight the real virus if you’re exposed in the future.
Why Herd Immunity Matters
Getting vaccinated doesn’t just protect you – it also helps protect your family, neighbors and community.
This wider protection is called herd immunity. It happens when enough people are immune to a disease (through vaccination or prior infection), making it harder for the disease to spread.
Herd immunity is especially important for protecting vulnerable people who can’t be vaccinated, such as newborns, elderly people, people with certain medical conditions or those undergoing cancer treatment.
The more people who are vaccinated, the stronger the community’s shield against outbreaks.
Booster Shots
Some vaccines require booster doses to maintain strong immunity. A booster “reminds” the immune system about the disease it learned to fight, strengthening protection over time.
For example, tetanus shots require boosters every 10 years. OVID-19 and flu vaccines are updated each year to include the most popular strains of the virus for that year.
Following your provider’s recommendations on boosters ensures your immunity stays strong.
Stay Up to Date on Vaccinations
Vaccines are a safe, effective way to protect yourself and others from serious illness. Staying current with recommended vaccinations is one of the best steps you can take to support lifelong health.
Talk to your primary care provider about:
- Which vaccines you or your children may need.
- Whether it’s time for a booster shot.
- How your vaccinations help build herd immunity in your community.
By getting vaccinated, you’re not just protecting yourself – you’re contributing to healthier, safer communities for everyone.